What General Conclusions Can You Draw About The Solubility Of Alcohols In Water
Chapter 11. Solutions and Colloids
xi.i The Dissolution Procedure
Learning Objectives
By the end of this section, you volition be able to:
- Describe the basic properties of solutions and how they form
- Predict whether a given mixture will yield a solution based on molecular properties of its components
- Explain why some solutions either produce or absorb heat when they course
An earlier affiliate of this text introduced solutions, defined as homogeneous mixtures of ii or more substances. Often, one component of a solution is nowadays at a significantly greater concentration, in which case it is called the solvent. The other components of the solution nowadays in relatively lesser concentrations are called solutes. Sugar is a covalent solid composed of sucrose molecules, C12H22O11. When this chemical compound dissolves in water, its molecules get uniformly distributed amidst the molecules of water:
[latex]\text{C}_{12}\text{H}_{22}\text{O}_{xi}(south)\;{\longrightarrow}\;\text{C}_{12}\text{H}_{22}\text{O}_{eleven}(aq)[/latex]
The subscript "aq" in the equation signifies that the sucrose molecules are solutes and are therefore individually dispersed throughout the aqueous solution (water is the solvent). Although sucrose molecules are heavier than h2o molecules, they remain dispersed throughout the solution; gravity does not cause them to "settle out" over time.
Potassium dichromate, Thou2Cr2Oseven, is an ionic compound composed of colorless potassium ions, K+, and orange dichromate ions, [latex]\text{Cr}_2\text{O}_7^{\;\;ii-}[/latex]. When a small amount of solid potassium dichromate is added to h2o, the chemical compound dissolves and dissociates to yield potassium ions and dichromate ions uniformly distributed throughout the mixture (Figure 1), as indicated in this equation:
[latex]\text{Grand}_2\text{Cr}_2\text{O}_7(s)\;{\longrightarrow}\;2\text{K}^{+}(aq)\;+\;\text{Cr}_2\text{O}_7^{\;\;ii-}(aq)[/latex]
As for the mixture of sugar and h2o, this mixture is too an aqueous solution. Its solutes, potassium and dichromate ions, remain individually dispersed amongst the solvent (water) molecules.


Visit this virtual lab to view simulations of the dissolution of common covalent and ionic substances (sugar and salt) in h2o.
Water is used then often as a solvent that the word solution has come up to imply an aqueous solution to many people. However, virtually any gas, liquid, or solid can act as a solvent. Many alloys are solid solutions of 1 metal dissolved in some other; for example, US v-cent coins contain nickel dissolved in copper. Air is a gaseous solution, a homogeneous mixture of nitrogen, oxygen, and several other gases. Oxygen (a gas), alcohol (a liquid), and sugar (a solid) all dissolve in water (a liquid) to form liquid solutions. Table 1 gives examples of several different solutions and the phases of the solutes and solvents.
| Solution | Solute | Solvent |
|---|---|---|
| air | O2(g) | Northward2(g) |
| soft drinks[i] | COii(yard) | HiiO(fifty) |
| hydrogen in palladium | Hii(yard) | Pd(s) |
| rubbing alcohol | H2O( fifty ) | C3H8O( l ) (two-propanol) |
| saltwater | NaCl( s ) | HtwoO( fifty ) |
| contumely | Zn( s ) | Cu( due south ) |
| Tabular array 1. Different Types of Solutions | ||
Solutions exhibit these defining traits:
- They are homogeneous; that is, afterwards a solution is mixed, it has the same composition at all points throughout (its composition is uniform).
- The concrete state of a solution—solid, liquid, or gas—is typically the aforementioned as that of the solvent, as demonstrated by the examples in Tabular array 1.
- The components of a solution are dispersed on a molecular scale; that is, they consist of a mixture of separated molecules, atoms, and/or ions.
- The dissolved solute in a solution will not settle out or separate from the solvent.
- The composition of a solution, or the concentrations of its components, can be varied continuously, inside limits.
The Formation of Solutions
The germination of a solution is an example of a spontaneous process, a process that occurs under specified weather condition without the requirement of free energy from some external source. Sometimes nosotros stir a mixture to speed up the dissolution procedure, but this is not necessary; a homogeneous solution would course if we waited long enough. The topic of spontaneity is critically important to the study of chemical thermodynamics and is treated more than thoroughly in a afterward chapter of this text. For purposes of this chapter'southward discussion, it will suffice to consider 2 criteria that favor, simply do not guarantee, the spontaneous germination of a solution:
- a decrease in the internal energy of the arrangement (an exothermic change, as discussed in the previous chapter on thermochemistry)
- an increment in the disorder in the system (which indicates an increase in the entropy of the system, as yous volition learn near in the later affiliate on thermodynamics)
In the process of dissolution, an internal free energy change oft, but non ever, occurs every bit rut is absorbed or evolved. An increase in disorder ever results when a solution forms.
When the strengths of the intermolecular forces of attraction between solute and solvent species in a solution are no different than those nowadays in the separated components, the solution is formed with no accompanying energy change. Such a solution is called an ideal solution. A mixture of ideal gases (or gases such equally helium and argon, which closely approach ideal behavior) is an example of an ideal solution, since the entities comprising these gases experience no significant intermolecular attractions.
When containers of helium and argon are connected, the gases spontaneously mix due to diffusion and form a solution (Effigy 2). The formation of this solution clearly involves an increase in disorder, since the helium and argon atoms occupy a volume twice equally large every bit that which each occupied earlier mixing.
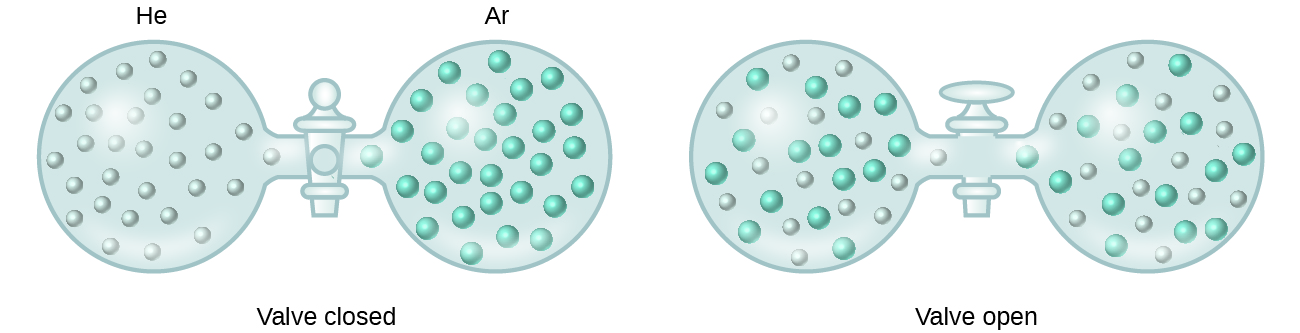
Ideal solutions may also form when structurally like liquids are mixed. For example, mixtures of the alcohols methanol (CH3OH) and ethanol (C2H5OH) course ideal solutions, as do mixtures of the hydrocarbons pentane, C5H12, and hexane, CviH14. Placing methanol and ethanol, or pentane and hexane, in the bulbs shown in Figure 2 will upshot in the same diffusion and subsequent mixing of these liquids equally is observed for the He and Ar gases (although at a much slower rate), yielding solutions with no significant change in energy. Dissimilar a mixture of gases, however, the components of these liquid-liquid solutions do, indeed, experience intermolecular bonny forces. Only since the molecules of the ii substances beingness mixed are structurally very like, the intermolecular attractive forces between similar and unlike molecules are essentially the aforementioned, and the dissolution process, therefore, does non entail any appreciable increase or decrease in energy. These examples illustrate how diffusion lone can provide the driving force required to crusade the spontaneous germination of a solution. In some cases, however, the relative magnitudes of intermolecular forces of attraction between solute and solvent species may prevent dissolution.
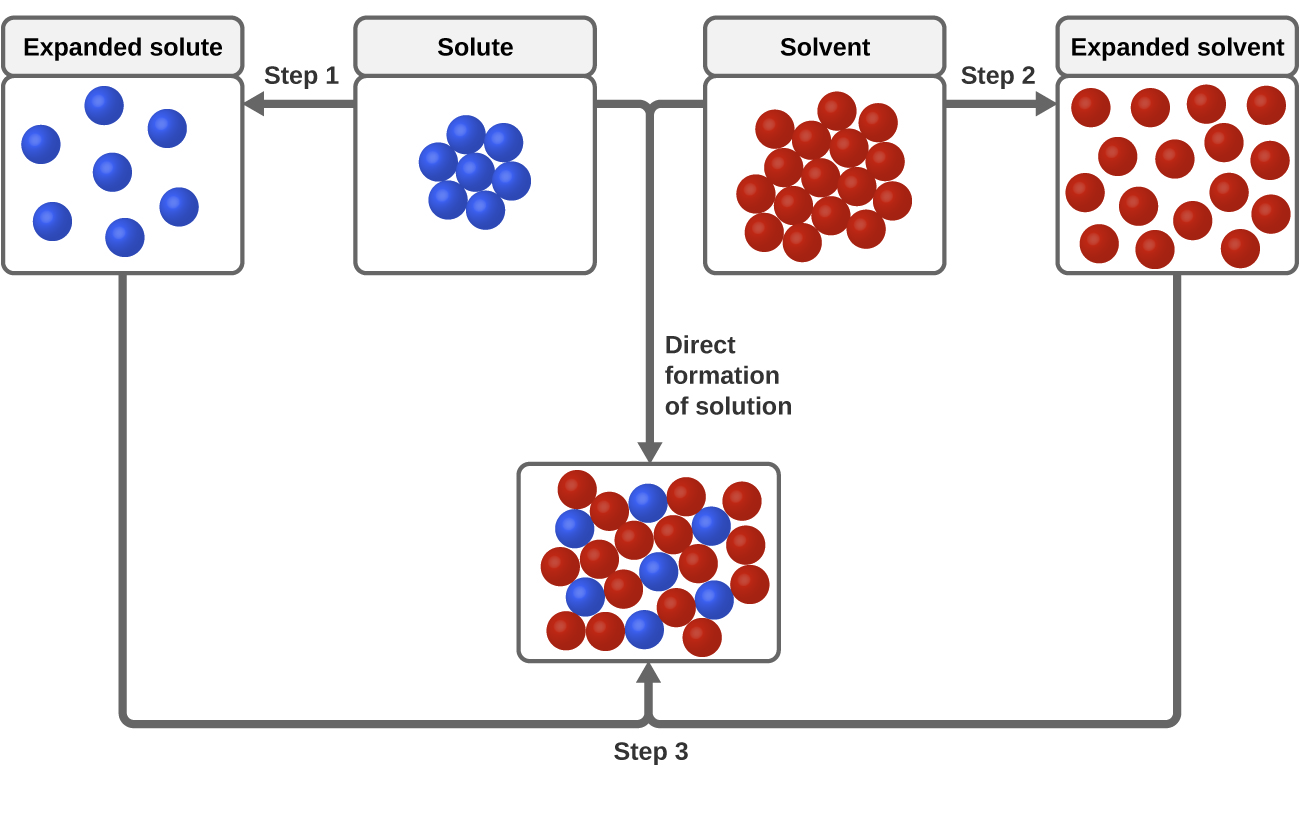
3 types of intermolecular attractive forces are relevant to the dissolution process: solute-solute, solvent-solvent, and solute-solvent. As illustrated in Figure three, the formation of a solution may be viewed as a stepwise procedure in which energy is consumed to overcome solute-solute and solvent-solvent attractions (endothermic processes) and released when solute-solvent attractions are established (an exothermic process referred to as solvation). The relative magnitudes of the free energy changes associated with these stepwise processes decide whether the dissolution process overall will release or absorb energy. In some cases, solutions do non course because the free energy required to separate solute and solvent species is so much greater than the energy released past solvation.
For example, cooking oils and h2o will not mix to any appreciable extent to yield solutions (Figure four). Hydrogen bonding is the dominant intermolecular bonny forcefulness present in liquid water; the nonpolar hydrocarbon molecules of cooking oils are not capable of hydrogen bonding, instead being held together by dispersion forces. Forming an oil-water solution would require overcoming the very strong hydrogen bonding in water, as well as the significantly strong dispersion forces between the relatively large oil molecules. And, since the polar water molecules and nonpolar oil molecules would not experience very strong intermolecular allure, very little energy would be released by solvation.

On the other hand, a mixture of ethanol and water will mix in whatever proportions to yield a solution. In this case, both substances are capable of hydrogen bonding, and so the solvation procedure is sufficiently exothermic to compensate for the endothermic separations of solute and solvent molecules.
As noted at the beginning of this module, spontaneous solution formation is favored, but not guaranteed, by exothermic dissolution processes. While many soluble compounds practice, indeed, deliquesce with the release of heat, some dissolve endothermically. Ammonium nitrate (NH4NOiii) is 1 such case and is used to make instant cold packs for treating injuries similar the one pictured in Figure 5. A thin-walled plastic purse of water is sealed inside a larger bag with solid NHivNO3. When the smaller pocketbook is broken, a solution of NHfourNOiii forms, absorbing oestrus from the surroundings (the injured area to which the pack is applied) and providing a common cold compress that decreases swelling. Endothermic dissolutions such equally this ane require a greater energy input to separate the solute species than is recovered when the solutes are solvated, but they are spontaneous nonetheless due to the increase in disorder that accompanies formation of the solution.


Watch this brief video illustrating endothermic and exothermic dissolution processes.
Key Concepts and Summary
A solution forms when ii or more than substances combine physically to yield a mixture that is homogeneous at the molecular level. The solvent is the nearly concentrated component and determines the physical state of the solution. The solutes are the other components typically present at concentrations less than that of the solvent. Solutions may grade endothermically or exothermically, depending upon the relative magnitudes of solute and solvent intermolecular attractive forces. Ideal solutions form with no appreciable change in energy.
Chemistry End of Chapter Exercises
- How exercise solutions differ from compounds? From other mixtures?
- Which of the principal characteristics of solutions can we see in the solutions of KtwoCr2O7 shown in Figure 1?
- When KNO3 is dissolved in water, the resulting solution is significantly colder than the water was originally.
(a) Is the dissolution of KNOiii an endothermic or an exothermic process?
(b) What conclusions can you draw virtually the intermolecular attractions involved in the process?
(c) Is the resulting solution an ideal solution?
- Give an example of each of the following types of solutions:
(a) a gas in a liquid
(b) a gas in a gas
(c) a solid in a solid
- Indicate the most important types of intermolecular attractions in each of the following solutions:
(a) The solution in Figure 1.
(b) NO(l) in CO(l)
(c) Cl2(g) in Br2(l)
(d) HCl(aq) in benzene C6Hsix(l)
(due east) Methanol CH3OH(l) in H2O(fifty)
- Predict whether each of the post-obit substances would be more soluble in water (polar solvent) or in a hydrocarbon such equally heptane (CsevenHxvi, nonpolar solvent):
(a) vegetable oil (nonpolar)
(b) isopropyl alcohol (polar)
(c) potassium bromide (ionic)
- Heat is released when some solutions grade; heat is absorbed when other solutions course. Provide a molecular explanation for the difference betwixt these 2 types of spontaneous processes.
- Solutions of hydrogen in palladium may be formed by exposing Pd metal to H2 gas. The concentration of hydrogen in the palladium depends on the force per unit area of H2 gas applied, but in a more than complex fashion than can exist described by Henry's police. Under certain weather, 0.94 g of hydrogen gas is dissolved in 215 g of palladium metal.
(a) Determine the molarity of this solution (solution density = ane.viii g/cm3).
(b) Make up one's mind the molality of this solution (solution density = 1.8 k/cm3).
(c) Determine the percent by mass of hydrogen atoms in this solution (solution density = 1.viii g/cm3).
Glossary
- blend
- solid mixture of a metallic element and one or more additional elements
- platonic solution
- solution that forms with no accompanying energy change
- solvation
- exothermic process in which intermolecular attractive forces betwixt the solute and solvent in a solution are established
- spontaneous procedure
- physical or chemical modify that occurs without the addition of energy from an external source
Solutions
Answers to Chemistry End of Chapter Exercises
1. A solution can vary in composition, while a compound cannot vary in composition. Solutions are homogeneous at the molecular level, while other mixtures are heterogeneous.
3. (a) The process is endothermic as the solution is consuming heat. (b) Attraction betwixt the K+ and [latex]\text{NO}_3^{\;\;-}[/latex] ions is stronger than between the ions and water molecules (the ion-ion interactions accept a lower, more than negative energy). Therefore, the dissolution procedure increases the energy of the molecular interactions, and it consumes the thermal energy of the solution to make up for the difference. (c) No, an ideal solution is formed with no appreciable heat release or consumption.
v. (a) ion-dipole forces; (b) dipole-dipole forces; (c) dispersion forces; (d) dispersion forces; (east) hydrogen bonding
7. Heat is released when the total intermolecular forces (IMFs) between the solute and solvent molecules are stronger than the total IMFs in the pure solute and in the pure solvent: Breaking weaker IMFs and forming stronger IMFs releases rut. Heat is absorbed when the total IMFs in the solution are weaker than the total of those in the pure solute and in the pure solvent: Breaking stronger IMFs and forming weaker IMFs absorbs heat.
Source: https://opentextbc.ca/chemistry/chapter/11-1-the-dissolution-process/
Posted by: santiagowareatur.blogspot.com


0 Response to "What General Conclusions Can You Draw About The Solubility Of Alcohols In Water"
Post a Comment